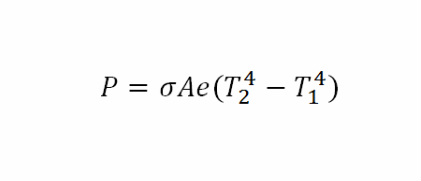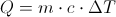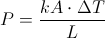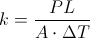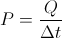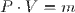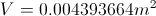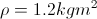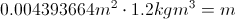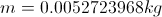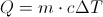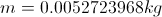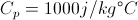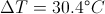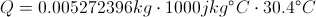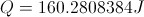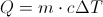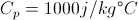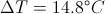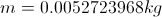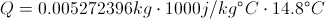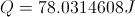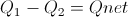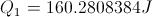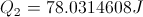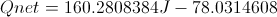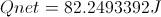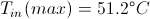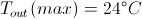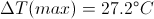First Law of ThermodynamicsThe total energy of an isolated system is constant dispite internal changes.
|
Second Law of ThermodynamicsEntropy of an isolated system never decreases.
|
Zeroth's Law of ThermodynamicsThe law that if two bodies are in thermal equilibrium with a third body then the first two bodies are in thermal equilibrium with each other.
|
Stefan's Law
- P = Rate of heat transfer
- σ = Stefans constant of 5.6696*10^-8
- A = Area of thermal conductivity
- e = Emisivity constant
- T = Temperature
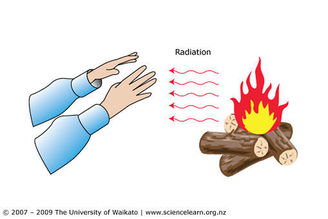
Radiation
The transfer of thermal energy through space by electro-magnetic waves.
R-ValueThe resistance of heat flow.
|
U-FactorThe coefficient of heat conductivity.
|
Variables
Q = Thermal Energy
m= mass
c = Heat Capacity
P = Rate of Heat Transfer
Delta t = Time dispacement
k = Thermal Conductivity
A = Area of Thermal Conductivity ( Measured in Meters )
L = Thickness of Thermal Conductor ( Measured in Meters )
Delta T = The Change in Temperature
m= mass
c = Heat Capacity
P = Rate of Heat Transfer
Delta t = Time dispacement
k = Thermal Conductivity
A = Area of Thermal Conductivity ( Measured in Meters )
L = Thickness of Thermal Conductor ( Measured in Meters )
Delta T = The Change in Temperature
Equations
This equation is for finding the thermal energy of something when you know the mass, heat capacity and the change in temperature. This equation can also be uses backward to find one of the variables on the inside when you know the thermal energy.
This equation is for finding the rate of heat transfer when you know the change in temperature, thermal conductivity, area of thermal conductivity and the thickness of thermal conductivity. This equation can also be used backward to find one of the variable on the inside when you know the rate of heat transfer.
This equation is for finding the thermal conductivity when you know the rate of heat transfer, the thickness of the object, the area of thermal conductivity, and the change in temperature. This equation can be used to find any one of the variables inside when you know the thermal conductivity.
This equation is for finding the rate of heat transfer when you know the thermal energy and change in time. You can use this equation backwards to find any one of the variable in it when you know the rate of heat transfer.
Lab Problem
The largest amount of energy consumed within the average home is related to maintaining adequate climate control through heating and cooling systems. To conserve energy and decrease expenses associated with climate control, proper home insulation techniques are required. Insulation technologies relating to materials and application have advanced throughout the home building industry with time. The home building industry once relied on straw and newspaper for insulating material. The industry currently utilizes technologies technologies such as fiberglass and blown expandable foam. Insulation material advancement is driven by consumers demanding insulation material designed for high insulation value along with positive new "green" homes. To meet the needs of green consumers, insulation manufacturers are developing insulation materials made from recycled products such as jeans, t-shirts and other low volatile organic products that can be treated with boric acid. Manufacturers have found that many green materials have other benefits to the homeowner as well, such as cotton's ability to provide excellent soundproofing.
- Your team will design a renewable composite insulation material.
Design Constraints
- Composite insulation material must produce minimum heat loss, representing good insulation.
- Composite insulation material must have overall uniform thickness less than or equal to one inch.
- Composite insulation material must have uniform internal composition.
- Individual insulation materials must be environmentally friendly.
- Individual insulation materials must be recyclable.
- Individual insulation materials must be economical.
- Composite insulation material dimension must not exceed the overall dimension of heat box apparatus.
- The change in temperature inside of the box is directly related to the heat absorbed or released by air in the box.
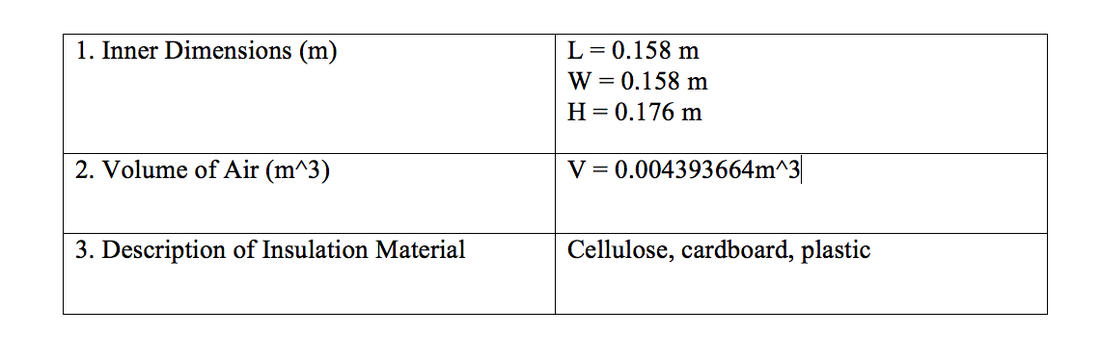
Materials
- I made cellulose for my insulation, and had a cardboard layer on the top and bottom to contain the the insulation. The cellulose is made of shredded paper and boric acid.
- Computer
- Heat box apparatus
- LoggerPro software
- 2 - Stainless steel temperature probes
- Tape
- Standard metric ruler
Procedure
- Plug two stainless steel temperature probes into two Go!Link connectors. Plug the Go!Link into one of the computers USB ports.
- Insert a temperature senor with acquisition capabilities inside the acrylic top cover of the heat transfer apparatus.
- Place or attach your fabricated composite insulation material to the outside top surface of the heat transfer apparatus.
- Place a second temperature sensor with acquisition capabilities outside of the box to measure the ambient room temperature.
- Open the program LoggerPro on your computer by clicking Start, All Programs, Vernier Software, LoggerPro 3.8.
- Activate heat source and leave on for 20 minutes or until the inside temperature remains relatively constant.
- While tester is heating up, set up LoggerPro to take data for 20 minutes.
- Click experiment, data collection.
- Change the experiment length to 20 minutes and the sample rate to 4 times per minute.
- Click OK
- Turn the heat source off and collect data for 20 minutes. To do so, click on the green Collect Data icon at the top of LoggerPro program.
- Once all of your data is collected, save the LoggerPro document.
- Remove temperature probes from heat box.
- Return the temperature probes and the Go!Link connectors to your teacher.
- You may obtain the necessary temperature data by examining the graph or using analysis tools in LoggerPro.
Pre-Test Data
Data Collected
Design Evaluation Calculations
All calculations are based on the combination of both your composite insulation material and the acrylic heat transfer apparatus material.
Calculate the Mass of the Air Being Heated
Select Equation
List All Known Values
Substitute Values Into Equation
Simplify and Solve for Mass
Calculate Energy Gained in Box During Heating
Select Equation
List all Known Values
Substitute Known Values Into Equation
Simplify and Solve for Energy Gained
Calculate Energy Lost During Cooling
Select Equation
List All Know Equations
Substitute Known Values Into Equation
Simplify and Solve for Energy Lost
Calculate Net Energy Retained in the Box
Select Equation
List All Known Values
Substitute Known Values Into Equation
Simplify and Solve for Net Energy Retained
Max In and Out
Conclusion
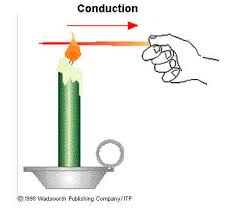
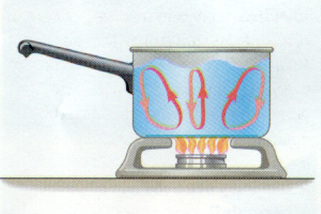
- Explain how your house might lose energy through radiation, convection and conduction.
- What modifications could be made to your team's insulation design that allow for more energy efficiency?
- Which beverage would be more beneficial for cooling you on a hot summer day: a cup of ice cold water or a cup of hot cocoa? Justify your choice.
- How do birds insulate their bodies to prevent energy loss on the skin's surface?
Birds use their feathers as insulation.
- Suppose that you are sitting close to a campfire. You decide to clean your glasses and you notice that your eyes feel warmer without your glasses. Explain the phenomenon.
Your eyes feel warmer because the radiation wouldn't have to go through the glass of your glasses but would go directly to your eyes and heat your eyes. Your glasses act as an insulation, yet a very bad one.
- We wear winter coats and cover with blankets to stay warm in the winter. If the coats and blankets are not a source of energy, how do we stay warm?
We stay warm by the thermal energy is trapped in by our insulation like the blanket and the cold is trapped out. Our body makes heat to keep us warm by homeostasis and that also keeps us warm.